
On April 28, 2026, the International Degree Program in Animal Vaccine Technology (IAVT) at NPUST hosted an insightful special lecture focusing on national regulatory standards for veterinary biologics. The invited guest speaker was I-Ting Ko, an Associate Researcher, Department Head, and Doctor of Veterinary Medicine (DVM). She represents the Animal Drugs Inspection Branch of the Veterinary Research Institute (VRI) under the Ministry of Agriculture. Her comprehensive presentation was titled “Animal Vaccine Management and Quality Control in Taiwan”


Dr. Ko introduced the attendees to the National Animal Vaccine Inspection Laboratory, highlighting its advanced infrastructure, which includes BSL-2 and ABSL-2+ facilities designated for biologics and animal testing. She emphasized that the laboratory operates under a stringent Quality Assurance System and holds the ISO/IEC 17025:2017 Testing laboratory accreditation granted by the Taiwan Accreditation Foundation (TAF). The lecture also outlined the coordinated animal vaccine management system in Taiwan, detailing the collaborative efforts between the Animal and Plant Health Inspection Agency (APHIA), the VRI, and local county or city governments.


A major highlight of the session was the detailed explanation of the registration process for veterinary biologics and the rigorous batch-by-batch inspections required for licensed veterinary vaccines before they reach the market. Dr. Ko explained that the batch inspection process is meticulously managed through a Laboratory Information Management System (LIMS). The evaluation includes a wide array of mandatory tests, such as characteristic tests, sterility tests, safety tests, potency tests, and virus content tests. She noted that all these procedures strictly adhere to Taiwan’s “Test Standards of Veterinary Drugs,” which govern the testing items for various vaccines, including those for swine, poultry, and aquatic species.
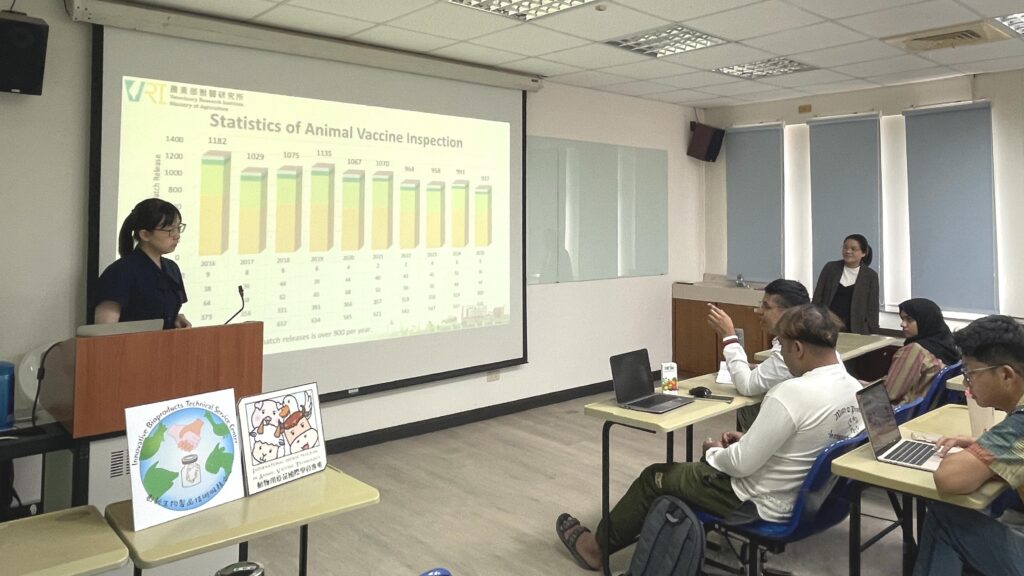
Furthermore, the speaker underscored the critical importance of post-marketing surveillance for veterinary biologics. This ongoing surveillance is designed to identify substandard veterinary products—such as those containing contaminated ingredients or exceeding their expiration dates—and to track down illegal veterinary products through routine audits and joint judicial investigations. To equip the audience with practical knowledge, Dr. Ko demonstrated how to properly identify approved vaccines by checking for the Official Release Seal affixed to each vial. The lecture provided IAVT students and researchers with invaluable regulatory perspectives, successfully bridging the gap between academic vaccine development and national quality control standards.


